High-throughput screening for cytotoxic T lymphocyte
inhibitors and analysis of their mechanisms of action
Background
Immune suppression is crucial in treating autoimmune diseases and to prevent transplant rejection. Current immunosuppressive drugs can cause multiple side effects such as infection and malignancy, and they do not prevent chronic transplant rejection. Therefore there is a great need to discover novel immunosuppressive compounds for therapeutic applications.
This project aims to screen compound libraries in order to identify potential immunosuppressive chemicals as pharmaceutical leads.

Method
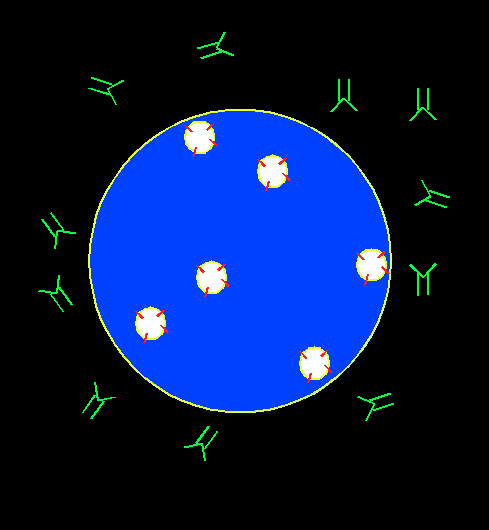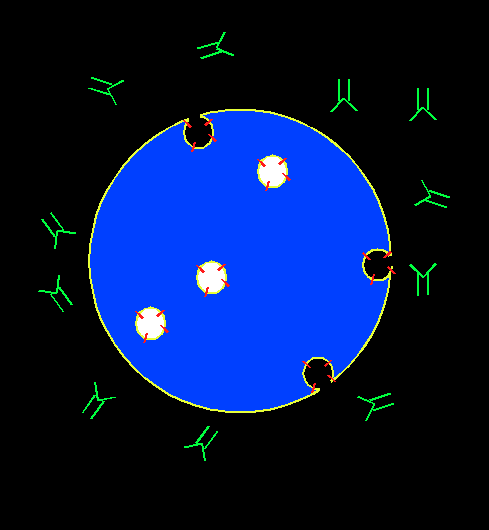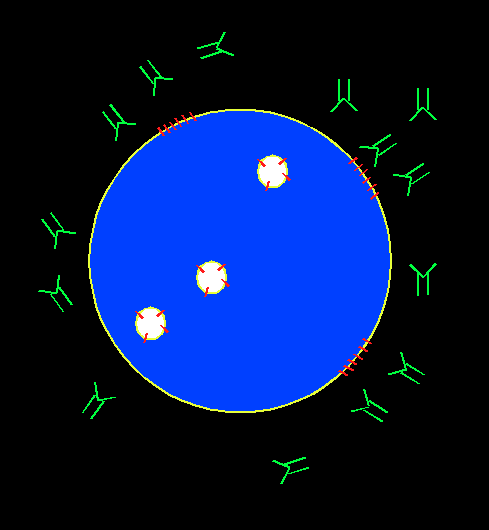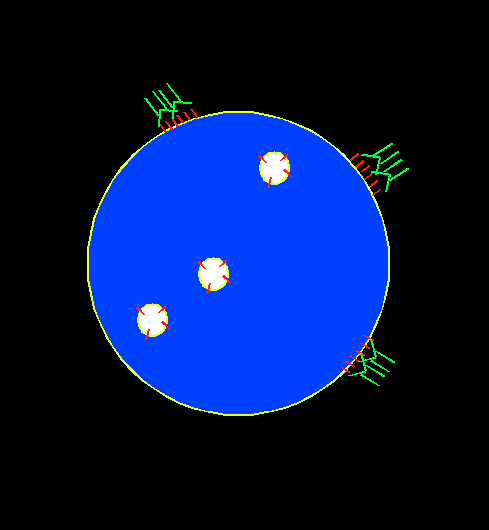
In order to screen for immunosuppressive chemicals, cytotoxic T lymphocyte (CTL) is chosen as a cell model for phenotypic screen. CTL can recognize and kill target cells such as tumor cells, virus infected cells, and non-self cells. It plays an important role in transplant rejection. The mechanism for its immediate cytotoxicity is lytic granule exocytosis, which releases cytotoxic agents such as granzyme and perforin that cause the target cell to lyse. The level for exocytosis can be detected through the binding of a fluorescent antibody to the lysosome-associated membrane protein (LAMP), which gets externalized to CTL cell membrane upon cell activation. The presence of an immunosuppressant will suppress cell activation, decrease the exocytosis level, and hence yield lower fluorescence intensity associated with LAMP.
Our assay is a novel, ready-to-screen, phonotypic screening assay designed to look for immune regulators.
Result and Significance
Our phenotypic assay is performed in high-throughput format to screen the molecular library small molecule repository, a chemical library including 364,202 diverse compounds. This identified 2,404 initial hits, 161 confirmed hits, and 75 biologically relevant hits with reasonable dose response. These 75 hits are obtained and further tested for molecular mechanism of action (MMOA) analysis.
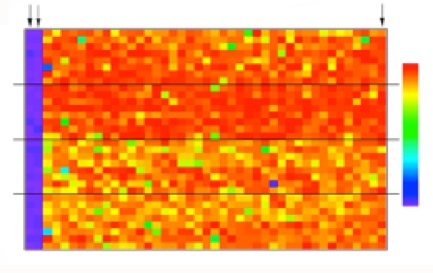
Heat map for a representative plate from the primary screening campaign

Flow chart showing the hit selection process from the primary screening.
Screening is conducted in collaboration with University of New Mexico Center for Molecular Discovery.
Immunosuppressant compounds identified in our screening may work by inhibiting one or more steps of the signaling network such as calcium influx, PKC and MKK activity, calmodulin and calcineurin activity, and ERK activity. They may also work by targeting novel regulatory molecules yet to be identified. Through MMOA analysis, effects of hits can be assigned to a known signaling event. The rest of the hits, without a known target, may represent chemical probes to identify the novel regulators.
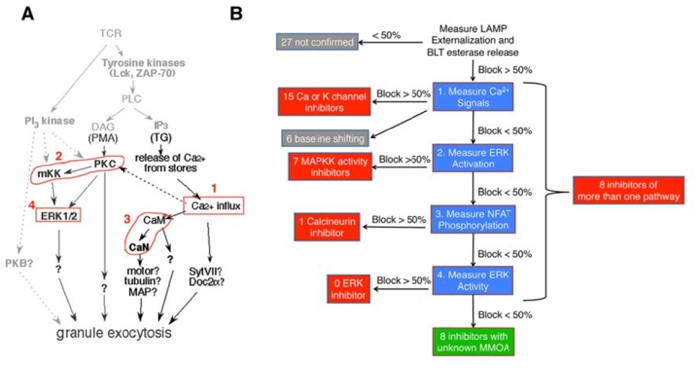
Since the signaling network leading to granule exocytosis in CTL overlaps extensively with that in helper T cells, inhibitory compounds identified in our screening may work as general immune inhibitors.
Future project
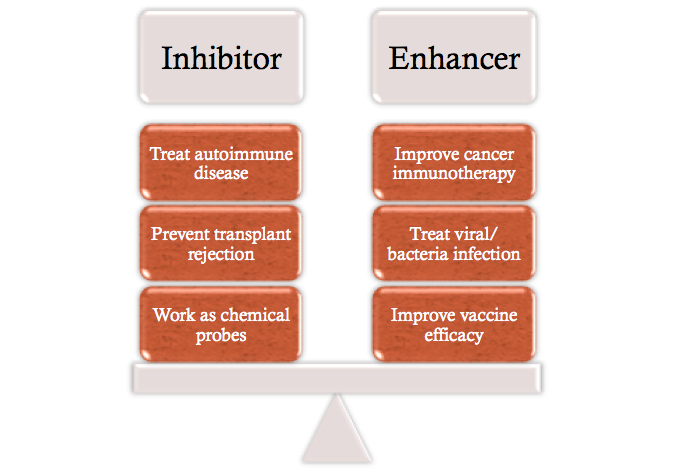
Currently there is an ongoing project to look for novel immune regulators using CTL exocytosis as a phenotypic screening assay. Instead of small molecules such as thapsigargin and phorbol myristate acetate, dynabeads coated with anti-CD3 are used as stimuli for CTL surface T cell receptor. This physiological level of stimulation enables the detection of regulatory molecules with either enhancing or inhibitory effects. The immune enhancing hits can be developed into therapeutics for tumor and cancer treatment, whereas the inhibitory hits can be used to treat autoimmune disease and to prevent transplant rejection.

This work was supported by R21 NS066462 to AZ and U54 MH084690 to LAS